Secondary Battery Materials
EPCHEMTECH supplies the top-quality electrolytes and additives.

Secondary Battery Materials: EPCHEMTECH supplies electrolytes and additives, which are materials for secondary batteries.
We develop and produce electrolytes and additives that enhance battery performance, including lifespan, stability, and charging speed.
Korea's first independent manufacturing technology for LiFSI in lithium-ion secondary batteries was developed in 2016,
and a patent for it was obtained. We continue to strengthen our technologies.
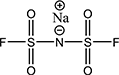
- Lithium salt
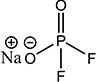
- Sodium Salt
- Additive
- Separator Binder
- Ionic Liquid Electrolyte
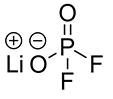
Lithium salt
TEL : Chemical Business Division 063-731-0045
Electrolyte
- Electrolyte plays a role in moving lithium ions between electrodes at battery charging and discharging
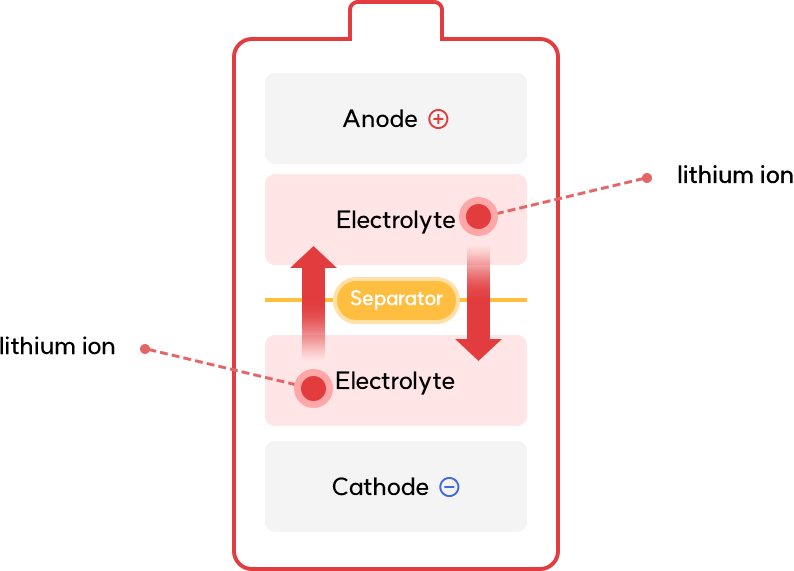
Next-generation Electrolyte
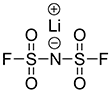
LiFSI
- Providing the highest iconic conductivity among electrolytes available in the market, producing excellent stability against high temperatures as the pyrolysis temperature is high
- Effects to increase battery lifespan, reduce charging time, and improve low-temperature performance verified
- The increasing amount of LiFSI electrolyte supplements the performance of the LFP battery
- Core electrolyte to be applied to new batteries such as lithium-sulfur battery
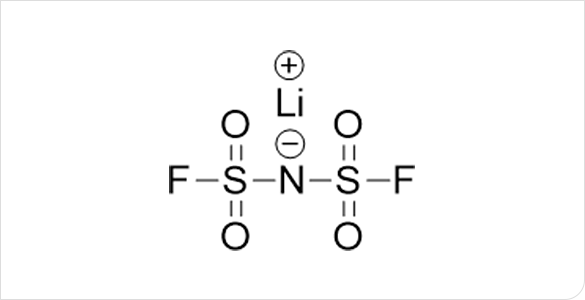

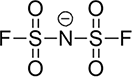
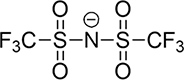
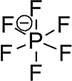
| Type | FSI | TFSI | PF6 |
|---|---|---|---|
 |
 |
 |
|
| Pyrolysis Temperature(°C) | 308 | 337 | 154 |
| Li+ Ionic Conductivity (10-3 S/cm) | 9.8 | 4.7 | 8.0 |
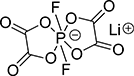
Sodium Salt
TEL : Chemical Business Division 063-731-0045
Additive
TEL : Chemical Business Division 063-731-0045
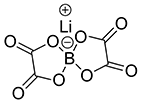
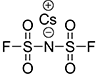
CsFSI
Additive to improve the stability of lithium-ion batteries against explosions, with a small amount added to the electrolyte
- Improving battery output at storage under high temperature
- Improving battery cycle life characteristics at ambient temperature and high temperature
- Effect to improve stability verified through battery penetration test
New additive supplied by EPCHEMTECH, output after storage under high temperature improved by 14% in comparison to VC addition
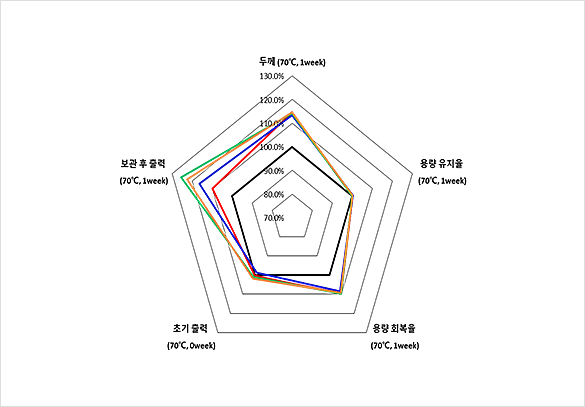
| Type | Electrolyte composition |
|---|---|
| LiPF6 in EC/EMC/DMC | |
| LiPF6 in EC/EMC/DMC + VC | |
| LiPF6 in EC/EMC/DMC + VC + LiFSI | |
| LiPF6 in EC/EMC/DMC + VC + CsFSI 0.5% | |
| LiPF6 in EC/EMC/DMC + VC + CsFSI 0.1% |
Separator Binder
| Name | Product Name | Features | Inquiry |
|---|---|---|---|
| EP-WB01 | Water-based binder 01 |
Prevent membrane shrinkage or rupture Explosion proof |
TEL : Chemical Business Division 063-731-0045
Separator Binder
- Core material to improve separator performance, strengthening safety by decreasing heat shrinkage rate
Water-Based Binder - EP-WB01
- Water based type thermosetting binder
- Excellent adhesion to ceramic materials (separator)
- Providing excellent heat resistance to prevent separator shrinking and improve explosion prevention performance
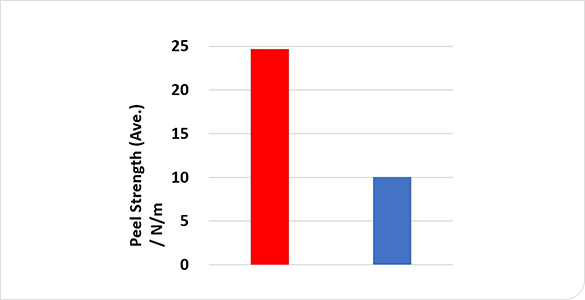
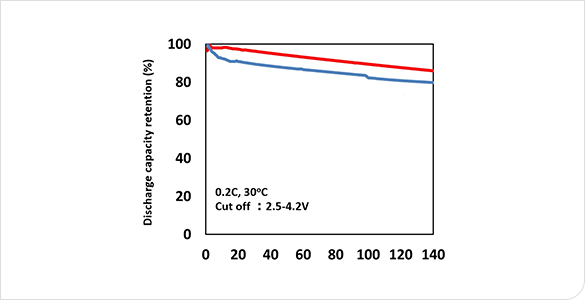
Comparison of adhesive strength and charge/discharge performance
Heat resistance test
Before heating

Coated separator

After heating in a 120 degree oven

Coated separator

Ionic Liquid Electrolyte
| Name | Product Name | Features | Inquiry |
|---|---|---|---|
| EP-IL01 | Ionic-liquid 01 |
Non-volatile, nonflammable Improving battery stability against explosion Preventing battery deterioration |
TEL : Chemical Business Division 063-731-0045
EP-IL01 (Ionic liquid electrolyte for solid-state battery)
Ionic liquid electrolyte containing FSI anion, used for flame-retardant products and solid-state electrolyte
- Non-volatile and nonflammable, used for flame-retardant products
- Self-extinguishing, improving battery stability against explosion
- Preventing battery deterioration by filling blank space between electrode and solid electrolyte
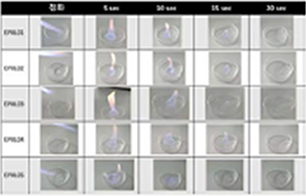
Self-extinguishing Features
Ionic liquid electrolytes have the characteristics of being non-flammable, can be mixed with organic solvents, and have the characteristic of
self-extinguishing in the event of a battery fire
At the initial stage of ignition, the fire is ignited by the carbonate solvent, which is a flammable organic solvent, but as time passes, self-extinguishment occurs by the liquid electrolyte, which is a flammable solvent.
- Polymer and solid electrolyte shorten the battery lifespan by forming pores (blank space) between the electrode and electrolyte
- Preventing battery performance deterioration and improving stability against explosion by filling pores using ionic liquid electrolyte as the solvent